Silver nitrate titration (the Mohr method) detects the amount of chlorine in a sample and converts it to salt concentration. ATAGO's Salt Meters use a concentration unit of weight/weight as a percentage, whereas silver nitrate titration (the Mohr method) uses weight/volume as a percentage. Due to these differences in principle and concentration unit, ATAGO's Salt Meters and silver nitrate titration (the Mohr method) may give different values when measuring the same sample.
There is a correlation between the two measurements, so it is useful to create a conversion chart for each sample type to compare the values produced by silver nitrate titration (the Mohr method) and ATAGO's Salt Meters.
Silver nitrate titration (the Mohr method and potentiometric titration) and salt meters using electrical conductivity use different methods of measurement. Especially for measurements of samples including substances other than NaCl, there is no consensus between the two measurements.
Silver nitrate titration has been popular for a long time as a method of measuring salt content, but it requires many instruments and a lot of time for preparations and cleanup. The equipment is complex, so measurements must be taken on a table, which means that the equipment cannot be taken on-site. A reagent (silver nitrate) is also necessary, creating a running cost. In addition, there are other drawbacks, including hassles after measurement such as disposal of waste liquid from the silver nitrate and the inability to perform measurements at food preparation sites due to the use of a reagent. ATAGO's Salt Meters using electrical conductivity do not require an apparatus or a reagent and can be used at food preparation sites, unlike silver nitrate titration. The Salt Meters can undergo a thorough washing, so there is no need to worry about sanitation.
For silver nitrate titration (the Mohr method or potentiometric titration), when measuring a solid sample, the unit is usually w/w (g/g), and when measuring a liquid sample, the unit is usually w/v (g/mL).
Additionally, the resulting measurements change depending on whether the sample undergoes preprocessing such as dissociation treatment of chlorine other than sodium chloride or dissociation treatment of bromine and iodine, which react to the silver nitrate. When measuring soy sauce or seawater, the salt concentration can be determined by multiplying by a coefficient. If there is a difference in the measurement from an ATAGO Salt Meter after performing this calculation, please check the measurement conditions for the silver nitrate titration.
ATAGO's Salt Meters (electrical conductivity) measure the sample's entire electrolyte composition. When there are components other than sodium chloride, the electrolyte's additional value is measured.
Therefore, if components other than sodium chloride are present in the sample, whether by silver nitrate titration or electrical conductivity, the components have an effect on the results of the measurement. Selecting the right tool for the job requires an understanding of both the advantages and drawbacks of each method.
When a sample where components other than sodium chloride are present is measured using silver nitration titration and the electrical conductivity method, the resulting measurements may differ, so in order to correlate these measurements we recommend creating a standard curve for each type of sample.
How to Create a Standard Curve
Here is an example of how to create a conversion graph.
Measure some samples with different salt content percentages (for example, condiments) using both silver nitrate titration (the Mohr method) and the ES-421/PAL-SALT.
For this example, when the Mohr method gave a result of 4.5% for a sample, the ES-421 gave a result of 4.2%. When the Mohr method gave 5.9%, the ES-421/PAL-SALT gave 5.5%. When the Mohr method gave 8.0%, the ES-421/PAL-SALT gave 7.0%, and so on... Plotted on the graph below, these results form a dotted line.
The conversion formula (line) created from these measurements is y = 1.24χ-0.78.
In this equation, χ represents the measurement from the ES-421/PAL-SALT, and y represents the measurement from the Mohr method.
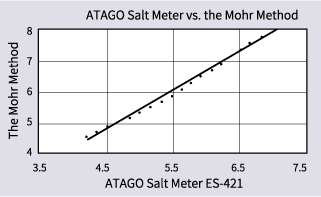
When correlating measurements taken using silver nitrate titration (the Mohr method) to measurements from ATAGO Salt Meters, use either a graph or a conversion formula like the example ones.
ATAGO Salt Meters are equipped with an offset function that can directly display the corrected value.
Further details about functions can be found in the instruction manual.

