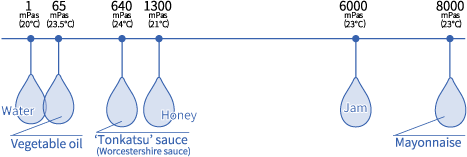
Жидкости могут быть свободно текущими, как вода, или липкими, как мед, а вязкость показывает, насколько «свободно течет» или насколько «липкая» жидкость.
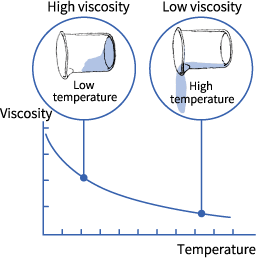
При повышении температуры вещества вязкость уменьшается. Например, все, наверное, уже сталкивались с тем, что при нагревании мед становится менее липким (менее вязким).
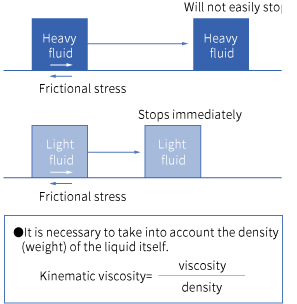
Вязкость показывает, насколько трудно объекту двигаться через жидкость. И наоборот, кинематическая вязкость показывает, насколько трудно самой жидкости двигаться. Вязкость представляет собой тормоза, наложенные на поток, но эффективность тормозов зависит от плотности жидкости. Это то же самое, как велосипед и автобус, движущиеся с одинаковой скоростью и при одинаковой силе торможения, все равно остановятся по-разному из-за разницы в весе. Кинематическая вязкость рассчитывается путем деления вязкости вещества на его плотность. Единицей кинематической вязкости является м2/с.